In 1911, he theorized that atoms have their charge concentrated in a very small nucleus. Together with Thomas Royds, Rutherford is credited with proving that alpha radiation is composed of helium nuclei. Rutherford's discoveries include the concept of radioactive half-life, the radioactive element radon, and the differentiation and naming of alpha and beta radiation. In 1908, he was awarded the Nobel Prize in Chemistry "for his investigations into the disintegration of the elements, and the chemistry of radioactive substances." He was the first Oceanian Nobel laureate, and the first to perform the awarded work in Canada. 
Rutherford has been described as "the father of nuclear physics", and "the greatest experimentalist since Michael Faraday". By the law of conservation of energy, an electron's kinetic energy should gradually decrease, leading to its spiralling motion into the nucleus.Ernest Rutherford, 1st Baron Rutherford of Nelson, OM, PRS, HonFRSE (30 August 1871 – 19 October 1937) was a New Zealand physicist who was a pioneering researcher in both atomic and nuclear physics. However, by Maxwell's electromagnetic theory, electrons should emit radiation when they experience centripetal acceleration. Rutherford proposed that electrons must be orbiting the positively charged nucleus like how satellites orbit the Earth, otherwise they would clash into the nucleus due to the electrostatic attraction towards the nucleus. The model fails to explain the stability of electrons' orbital motion. There are three main limitations to Rutherford's atomic model:ġ. Limitations of Rutherford's Model of the Atom 
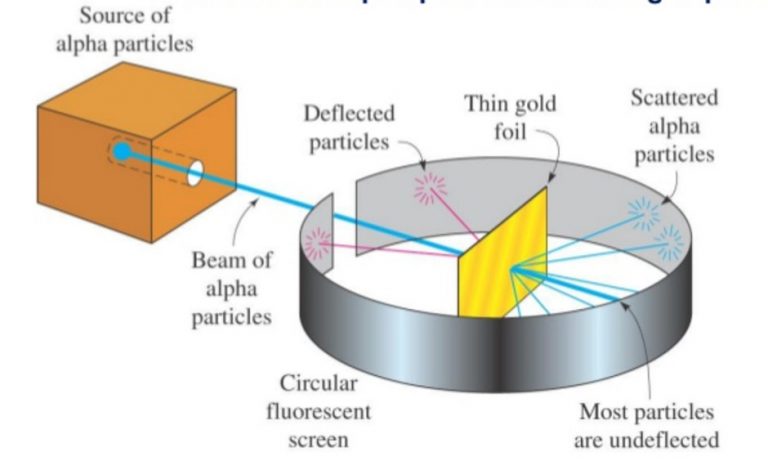
He showed that a proton is positively charged and much heavier than an electron.Īlthough Rutherford demonstrated that the charge of proton(s) accounts for the positive nature of the nucleus, he wasn't able to account for the nuclear mass. Rutherford conducted a similar experiment as Thomson to determine the value of the charge to mass ratio of a proton. Rutherford fired alpha particles at a sample of nitrogen gas, which resulted in a transmutation reaction producing protons. How was the Proton Discovered?Īfter Geiger and Marsden's gold foil experiment, Rutherford tried to investigate the content of the nucleus. Geiger and Marsden's gold foil experiment not only provided evidence for Rutherford's model of the atom, it rejected the preceding atomic model proposed by Thomson. Although Thomson's model predicted that all, if not most, alpha particles would pass through the gold foil undeflected, it could not account for the few alpha particles that were reflected. As a result, when an alpha particle collided with the nucleus, it was reflected. In an atom of gold, the charge and mass of the nucleus are substantially greater than that of an alpha particles. This observation supported the presence of a region in the atom of highly concentrated positive charge (nucleus). The angle of deflection was measured by the position at which they were detected on the fluorescent screen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
